|
18, 19 These agents include humanized IgG antibodies against programmed death 1/programmed death ligand 1 (PD-1/PD-L1) and cytotoxic T-lymphocyte-associated protein 4 (CTLA-4). 16, 17 The theoretical benefits of administering immunotherapy in the neoadjuvant setting include increased efficacy of antitumor immune activity in the setting of an intact (rather than resected) primary tumor that is capable of shedding neoantigens from dying tumor cells, thereby increasing T-cell priming and redirection and facilitating a stronger immune response. In the modern era, immunotherapy with immune checkpoint inhibitors (ICIs) has shifted the treatment paradigm of NSCLC in both the metastatic and resectable setting. 13, 14 However, the NSCLC Meta-analysis Collaborative Group published a meta-analysis in the Lancet in 2014 demonstrating similar improvement in OS with neoadjuvant chemotherapy to adjuvant therapy, 15 and up until recently, physicians were using the two interchangeably. 12 This ultimately led to a paucity of treatment recommendations in the neoadjuvant space, despite arguments that patients may have better performance status preoperatively and demonstrate increased compliance to treatment. 1įollowing the adoption of adjuvant cisplatin-based chemotherapy for NSCLC, several neoadjuvant studies closed due to accrual difficulties. 7 – 11 While the lack of standardization between patient selection and regimens initially led to both positive and negative results, the OS benefit of adjuvant cisplatin was later confirmed in the 1995 LACE meta-analysis, paving the way for widespread clinical use. In the interim, multiple large clinical trials had opened investigating adjuvant chemotherapy with systemic cisplatin-based regimens in resectable NSCLC. 3 This was quickly followed by multiple negative studies showing both a lack of overall survival (OS) benefit and potential harm, 4 – 6 which led to adjuvant PORT falling out of clinical favor. 1 Adjuvant therapy was first adopted by some practices in the 1980s in the form of postoperative RT (PORT) after some studies showed a potential benefit in local recurrence rates. 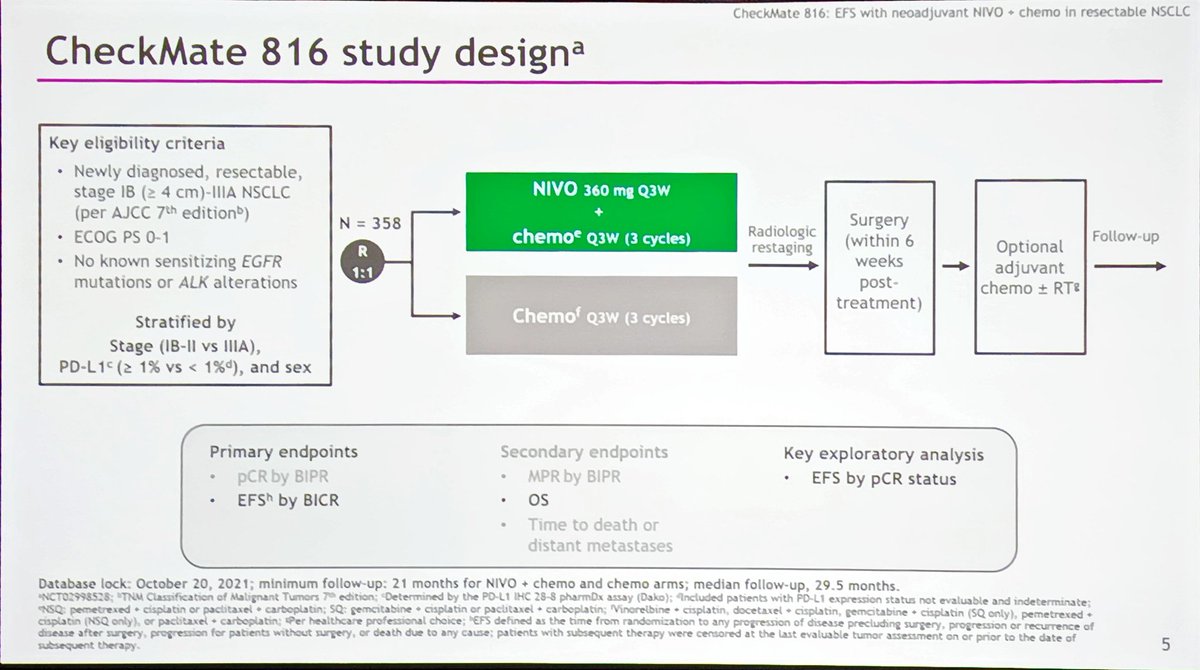
2 Stage IA patients are not typically recommended to receive adjuvant (or neoadjuvant) systemic treatment as the risks of therapy have been observed to outweigh any survival benefit in meta-analyses. Resectable NSCLC is generally defined as stage I–IIIA disease, utilizing American Joint Committee on Cancer (AJCC) 8th edition guidelines, but ultimately determined by the surgeon. Ultimately, the complexity of managing resectable NSCLC highlights the importance of a multidisciplinary approach to patient care.Īlthough adjuvant therapy has traditionally been a key component of resectable non-small-cell lung cancer (NSCLC), 1 the advent and efficacy of immunotherapy has provided an inevitable push to move treatment into the neoadjuvant setting. Longer follow-up of CheckMate-816 and other ongoing phase III trials may help address these questions. Despite the growing literature and success of these trials, several outstanding questions remain, including the relationship between pathologic response and patient survival, the role of biomarkers such as programmed death ligand 1 and circulating tumor DNA in determining patient selection and treatment course, and the utility of additional adjuvant therapies. CheckMate-816, which was a randomized phase III trial studying neoadjuvant nivolumab in addition to chemotherapy, definitively established a benefit for neoadjuvant chemoimmunotherapy compared to chemotherapy alone for resectable NSCLC. Across these trials, neoadjuvant chemoimmunotherapy led to high rates of pathologic response and improved surgical outcomes without compromising surgical timing or feasibility. Significant interest in neoadjuvant chemoimmunotherapy resulted in the conduct of multiple successful phase II trials including the Columbia trial, NADIM, SAKK 16/14, and NADIM II. The phase II LCMC3 and NEOSTAR trials demonstrated a role for neoadjuvant immunotherapy in inducing meaningful pathologic responses, and another phase II trial established the feasibility of combining neoadjuvant durvalumab with RT. A growing number of promising trials have examined the utility of neoadjuvant immunotherapy, both alone and in combination with other modalities such as radiation therapy (RT) and chemotherapy. 
The advent of immune checkpoint inhibition has pushed the treatment paradigm for resectable non-small-cell lung cancer (NSCLC) toward neoadjuvant therapy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
